studyDESIGNER simplifies GLP and non-GLP studies by managing data acquisition, data analysis, and reporting steps during toxicology and safety pharmacology studies.
studyDESIGNER’s modular platform builds and organizes the study with fully integrated data management, workflow guidance, and automation at each stage of the experimental process.
Automated post-processing capabilities shorten time to reporting. Built-in audit-trail and electronic signature capabilities ensure data integrity and accountability.
FOCUS ON WHAT’S IMPORTANT
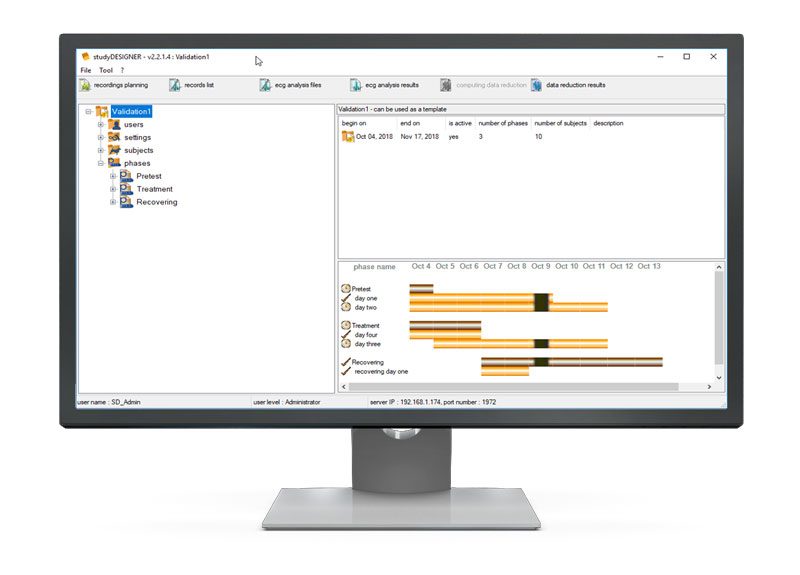
Users are guided in a stepwise manner through the various stages of the study – from study creation and study start through completion as well as archiving.
User level access provides granular control for system admins and shows users only what they are able to use and configure. This reduces complexity for the users that really need to focus on the animals and not the software.
The system is easily configured and designed for either long or short recordings from a few minutes to to multiple days.
During recording sessions studyDESIGNER automatically drives our embedded IOX2 (data acquisition) and ecgAUTO (pattern recognition post-processing) applications to record and analyze data and send calculated parameters to its database.
To comply with regulatory requirements, optional GLP modules can be activated on studyDESIGNER software. Data consistency, reliability and integrity are ensured by:
The usersMANAGEMENT module controls the access rights by login and password.
Access rights are characterized by a level (e.g. simple user, administrator) with an optional expiry date and a logout period.
auditCENTRAL software collects audit lines sent to Windows event logs of all client computers and keep them into a database. Each entry in the audit trail contains a list of all actions done on emka TECHNOLOGIES programs that affect or potentially affect raw or processed data, such as the date and time of the action, the user who performed the action or the computer on which the action was performed.
The eSIGN module offers encrypted electronic signatures.
studyMANAGEMENT module associates data files to a study.
